Notice of Class Certification and Settlement Approval
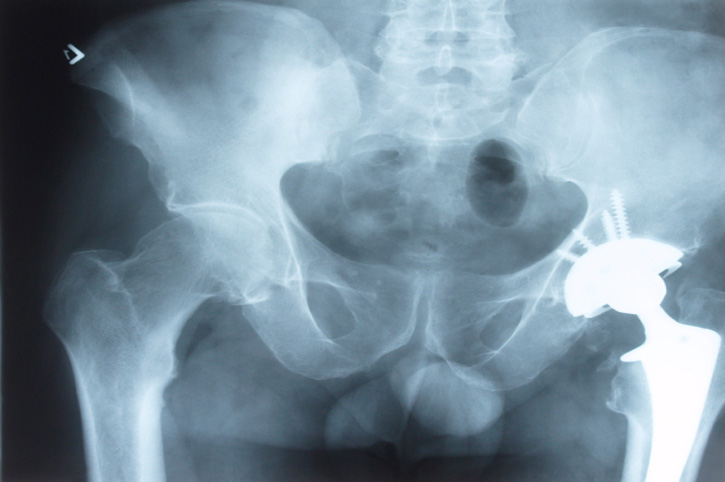
Klein Lawyers are class counsel in a class action lawsuit filed with the Supreme Court of British Columbia concerning individuals implanted with the DePuy ASR XL Acetabular Hip System and DePuy ASR Hip Resurfacing System. A Settlement Agreement has been signed by the parties on February 9, 2021 and approved by the Honourable Justice Ward Branch on February 26, 2021. A copy of this approval order is here.
For inquiries, kindly email dlennox@callkleinlawyers.com or dtanjuatco@callkleinlawyers.com.
“The negotiations were conducted by plaintiffs’ counsel with experience in class proceedings.”
“I am satisfied it is apparent from the material, which is complicated, and, I might say very well organized...”
“Plaintiff’s counsel are senior members of the bar who have extensive experience in personal injury litigation and class actions..”
“Mr. Klein is experienced and able in relation to class proceedings. Over time he has acquired expertise that permits him to make a valuable contribution...”
“The lead class counsel is experienced and has been recognized by the courts in approving settlements in other class actions.”
“Counsel for the plaintiffs in the various jurisdictions appear to be experienced in class proceedings, and to be recognized as skilled litigators.”
“…Plaintiff’s counsel retained the British Columbia law firm of Klein Lawyers to assist in the litigation. That firm has extensive experience...”
“Klein Lawyers is a litigation firm focusing on class actions. The firm is based in Vancouver but also has a Toronto office.”
“The proposed Class Counsel have excellent qualifications…”
“Mr. Klein of Klein Lawyers has over 20 years of experience in the field of class action litigation and has appeared as plaintiffs’ counsel in over 25 certified class actions...”
“Class counsel, Klein Lawyers LLP and Kim Orr Barristers P.C., are highly experienced in class action litigation. Both firms have practiced in the specialized area of class action litigation for over 20 years”
“British Columbia class counsel took on this case on a contingency basis, and faced a real risk of not being paid at all for their services.”
“Class counsel pursued this litigation to completion on their own rather than with a consortium of counsel from various provinces.”
“Klein Lawyers has continuously posted updates about the case on its website during the litigation, and has posted the court decisions...”
“There is little doubt that the lawyers of the firm expended a great deal of time in prosecuting the action, in negotiating a settlement...”
“…Class counsel undertook an extensive communication plan to advise potential class members of the proposed settlement and to advise them of the...”